Types of ATMPs
ATMPs represent a rapidly evolving area of modern medicine and require robust governance, specialist handling and multidisciplinary collaboration. These innovative medicines fall into three main categories:
- Gene therapy medicinal products
- Somatic cell therapy medicinal products
- Tissue engineered products
Gene therapy medicinal products (GTMPs)
GTMPs work by delivering genetic material into cells to regulate, repair, or replace faulty genes. They may be used for both therapeutic and diagnostic purposes.
These therapies introduce recombinant gene (segments of DNA assembled from different sources) into the body. Many GTMPs are classified as genetically modified organisms (GMOs) and therefore require additional governance and regulatory oversight.
However, not all GTMPs are considered GMOs. Examples include:
- some cancer immunotherapies using biologically produced mRNA technologies
- therapies using gene-editing platforms (for example CRISPR technologies)
Requirements for governance and preparation of gene therapy (SPS page) provides further detail on GTMP subtypes.
The handling and implementation of GTMPs differ depending on whether nucleic acid insertion occurs:
- in vivo – inside the patient’s body
- ex-vivo – outside the patient’s body as part of the manufacturing process
Pharmacy institutional readiness guidance is available for each GTMP type on the ATMP advice and guidance page (SPS page).
Somatic cell therapy medicinal products (sCTMPs)
sCTMPs are produced from cells or tissues that have been:
- substantially manipulated
- modified so their biological characteristics or functions differ from their natural physiological role.
These therapies aim to treat, cure, prevent or diagnose disease. Examples include tumour-infiltrating lymphocytes (TILs) and virus-specific cytotoxic T-lymphocyte (CTL) therapies.
Pharmacy institutional readiness guidance is available for sCTMPs on the ATMP advice and guidance page (SPS page).
Tissue engineered products (TEPs)
TEPs are medicines made from modified cells or tissues with the aim of repairing, replacing or regenerating human tissue. They often involve cells embedded within a biodegradable matrix or scaffold to restore structural or functional capacity.
Examples of tissue engineered products include autologous chondrocyte implantation and limbal stem cell transplantation.
The role of pharmacy
ATMPs are medicines, and the Chief Pharmacist holds organisational responsibility for their safe and compliant use. Pharmacy teams are central to their delivery, providing the governance and technical expertise needed for implementation.
Getting started with ATMPs
The Pan-UK Pharmacy Working Group for ATMPs provides national guidance to support safe and optimal ATMP implementation across the NHS.
Recommended steps for introducing the first ATMP into an organisation include:
1. Establish organisational governance and infrastructure
- Ensure appropriate clinical, pharmaceutical and financial expertise is in place to evaluate ATMP proposals. Guidance for Chief Pharmacists (SPS page) is available to support and guide initial ATMP implementation.
2. Assess each ATMP through formal governance
This includes:
- feasibility and regulatory assessment
- risk assessment of the product and patient journey
- an example GMO risk assessment is available in Appendix 2 of requirements for governance and preparation of gene therapy (SPS page)
- a preparation risk assessment tool to determine the optimal preparation location is available in performing preparation risk assessments for ATMPs (SPS page).
The Pan-UK Pharmacy Working Group for ATMPs has produced clinical area or pharmacy aseptic preparation worksheets for complex marketed ATMPs. Further information for individual marketed ATMPs including preparation risk assessments and worksheets is available via NHSFutures (NHS email required).
3. Undertake a gap analysis
- Use the relevant pharmacy institutional readiness guidance to identify gaps and ensure the full end-to-end process is documented appropriately.
4. Implement with SOPs and multidisciplinary collaboration
- Governance processes for onboarding should be clear for marketed, investigational and unlicensed ATMPs. For example, contracts with MA holders and sponsors will be required.
- Clear procedures, training and cross-speciality working are essential for safe delivery.
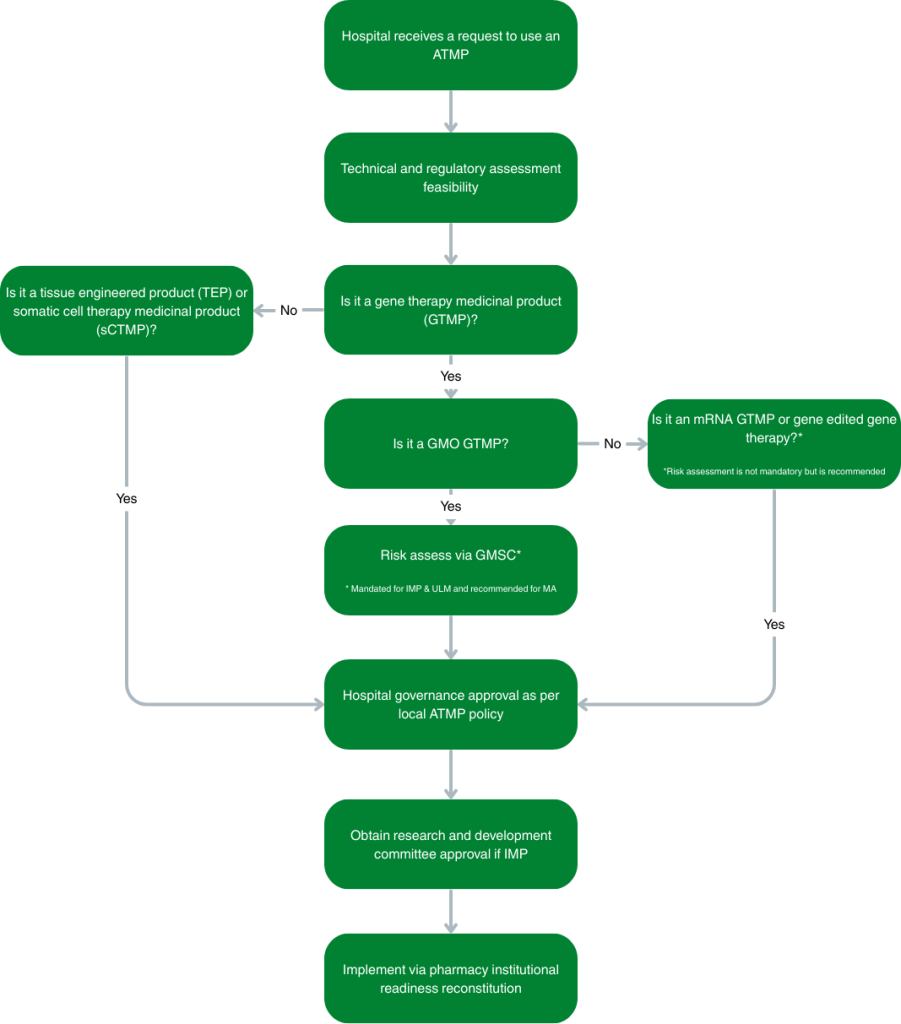
Organisational governance arrangements for initiating an ATMP
A recommended organisational governance process is outlined in the figure below: