Presentation
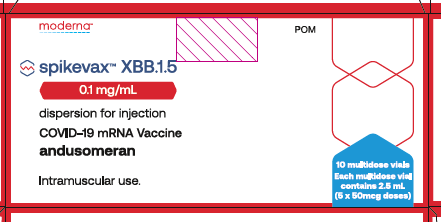
Spikevax (XBB.1.5) 0.1mg/mL dispersion for injection is presented in 2.5mL multi-dose vials in a ready to administer form. We refer to it as Spikevax (XBB.1.5) in our web articles and SOPs.
Spikevax (XBB.1.5) is thawed by the NHS’s Specialist Pharmaceutical Logistics (SPL) providers before onward supply to the NHS at 2-8°C. The SPLs supply whole thawed cartons each containing 10 multidose vials.
The thaw label applied by SPLs bears the new shelf life at 2-8°C. The vials inside the cartons are not labelled with the post thaw expiry date so it is essential that the vials remain within their carton until the point of removal from the fridge for immediate use.
Further information may be found on our Ordering, receipt & storage of COVID-19 Vaccines page.
Carton / Vial Artwork
Carton artwork
Vial artwork
Consumables and Patient Information Leaflets
Patient Information Leaflets, and syringes and needles for administration are provided with the vaccines.
Shelf life and storage
- Spikevax (XBB.1.5) may be stored for up to 30 days between 2 and 8°C following thawing.
- Within the 30 days, it may be stored for up to 24 hours at room temperature (between 8°C and 25°C) following removal from the refrigerator.
- Once punctured, the vaccine vial may be stored for up to 6 hours at room temperature up to 25°C, (within the allowed use period of 30 days between 2°C and 8°C and 24 hours between 8°C and 25°C). However, from a microbiological point of view, unless the method of puncture precludes the risk of microbial contamination, the product should be used as soon as practically possible.
Shelf life extensions
From time to time, variations to the marketing authorisation may mean that some batches may have shelf lives beyond the labelled manufacturer’s expiry date. If there are any applicable shelf life extensions, they will be listed on our vaccine expiry extension page.
Transport and movement
Unpunctured vaccine
Transit of unpunctured vials between 2 and 8°C can occur for a maximum of 30 hours by road. This may be broken into multiple separate journeys totalling no more than 30 hours.
It is assumed that 14 hours of transport time will already have been used during delivery to the NHS, which means that another 16 hours remain for transport once received by the NHS. The NHS logistics providers will inform the NHS if their journey has been longer than 14 hours.
Further information can be found on our Transport of COVID-19 vaccines article and our Mutual Aid guidance.
Punctured vaccine
The decision to move punctured vials between locations within the same legal entity must include an assessment of the risk of microbial contamination and proliferation versus risk of wastage and loss of opportunities to administer vaccines at alternative locations.
Any decision to move punctured vials must be made locally under the direction of the Chief Pharmacist or site lead pharmacist, taking the specific circumstances into account, and using appropriate risk control measures such as temperature control, infection prevention and control, and a means to identify that the vial has been punctured.
Preparation
Spikevax (XBB.1.5) is presented as a ‘ready to use’ formulation
Each dose is 0.5mL for individuals 12 years of age and older. See Spikevax (XBB.1.5) SPC for further dosing information.
Detailed advice about preparation, including a model standard operational procedure can be found on our Spikevax (XBB.1.5) preparation page.
Risk management
Spikevax (XBB.1.5) will be handled in the same location as other vaccines. Following the guidance on Handling multiple COVID-19 Vaccines will reduce the risk of errors.
Licensed status
Spikevax (XBB.1.5) has a Conditional Marketing Authorisation (PLGB) in the UK.
Allergy, excipients and dietary advice
Spikevax (XBB.1.5) is contraindicated in those with a hypersensitivity to the active substance or to any excipient (ingredient) in the vaccine, although revaccination may be possible on the advice of an expert (e.g. allergy specialist). See the Summary of Product Characteristics (SPC) for full details of the active substance and excipients included in the vaccine.
Allergy to any excipient is possible, but polyethylene glycol (PEG) is of particular importance. See the Green Book Chapter 14a: COVID-19 for details on how to manage individuals with a history of allergy.
Spikevax (XBB.1.5) is produced by cell-free invitro transcription. The SPC does not indicate presence of egg, gluten, nut, soy or latex. Further information can be requested directly from the manufacturer if required.
Useful resources
Refer to the following recommended resources for further information.
Summary of Product Characteristics
Spikevax (XBB.1.5) 0.1 mg/mL dispersion for injection
Update history
- Assumed time in transport by special logistics providers changed from 24 to 14 hours
- Reference to DHPC relating to transport removed as this information is now in the SPC
- Allowed time in transport updated in accordance with DHPC issued by Moderna
- Published